Monoclonal Antibody Therapy
Newman Regional Health provides COVID-19 monoclonal antibody therapy (mAb) to eligible patients in accordance with the Food & Drug Administration (FDA) Emergency Use Authorization (EUA) criteria. Monoclonal antibody therapy may be available to patients meeting specific criteria and with a provider order only.
What is Monoclonal Antibody therapy
Monoclonal antibody (mAb) therapy, also called monoclonal antibody infusion treatment, is a way of treating COVID-19. The goal of this therapy is to help prevent hospitalizations, reduce viral loads and lessen symptom severity.
Monoclonal antibodies are mass-produced in a laboratory, resemble your natural antibodies, and are designed to recognize the spike protein on your cell’s outer shell.
These specific antibodies interfere with the virus’s ability to attach and gain entry into human cells, thus assisting your natural immune system to mount its own response.
This therapy can be extremely effective but is not a replacement for vaccination.
Monoclonal antibodies, or mAbs, are made in a laboratory to fight a particular infection—in this case, SARS-CoV-2—and are given to patients directly with an infusion.
combatcovid.hhs.gov
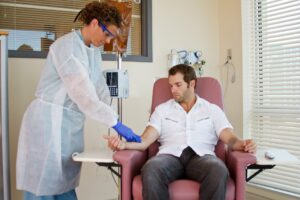
How is monoclonal antibody therapy administered?
Monoclonal antibody therapy is given through an intravenous (IV) under the direction of a medical professional.
These infusions are administered over about an hour, followed by an hour of observation and monitoring.